Antinol® Latest Studies
Sorted by recent
 Filter Studies by Smart Tags
Filter Studies by Smart Tags
Bone & JointCardiovascularCase Study & ProceedingCatDogEAB 277™EfficacyExotic PetImmunologyNeuro & BehaviorOphthalmologyOralRabbitRenalRespiratoryRodentSafetySkinUTI
Smart Search
Antinol® EAB-277® – The Executive Summary

Antinol® Rapid is a potent synergistic blend of 2 marine lipid extracts chosen for their unique enhancement formula called EAB-277®.
EAB-277® is the key active ingredient of this advanced formula formulated to promote optimal benefits through its synergistic efficacy contains > 90 free fatty acids full spectrum of Omega 3 including ETA, EPA, DHA as well as other key Polyunsaturated fatty acids (PUFAs) and antioxidants.
The two marine lipids used in Antinol® Rapid are proprietary and exclusively produced. The exact combination of 30mg lipid fractions from Perna canaliculus (New Zealand green lipped mussel) and 20mg high phospholipid krill oil is the result of years of research combining and isolating lipid groups and essential fatty acids to find the optimal nutrient synergy.
EAB-277®’s proprietary high phospholipid krill oil is high in polar lipid enrichment which enhances bioactivity “Potency” of this marine oil blend formula as a result of proven efficacy.
The Antinol® Rapid EAB-277® blend has been proven via laboratory tests to be more effective than either of the individual lipids alone in inhibiting inflammation markers such as nitric oxide, TNFα, and IL-6.



A randomised, double-blinded, controlled trial to determine the efficacy of combined therapy of oclacitinib and marine oil extract PCSO-524 in dogs with atopic dermatitis



Preliminary Study on Treatment Outcomes and Prednisolone Tapering after Marine Lipid Extract EAB-277 Supplementation in Dogs with Immune-Mediated Hemolytic Anemia


Simple Summary: Immune-mediated hemolytic anemia (IMHA) in dogs is a common autoimmune disease which is accompanied with a high death rate and therapeutic challenges. A natural antiinflammatory nutraceutical product, EAB-277, is derived from marine lipids. Unfortunately, the effects of EAB-277 in IMHA dogs has rarely been investigated. The objective of this study is to assess the clinical effects of EAB-277 and prednisolone dose-tapering for supplemental therapy in IMHA dogs. The findings provide evidence that standard therapy combined with EAB-277 can improve hematological and blood chemistry profiles, resulting in a higher survival rate in IMHA dogs. Furthermore, EAB-277 supplementation can reduce prednisolone dosage tapering and improve the quality of life of IMHA dogs. However, a longer-term study with a larger sample size is necessary to corroborate these findings. As a result, marine EAB-277 is a promising alternative to existing medication for IMHA. Since the nutraceuticals have been utilized not only for nutrition but also as supplemental therapy for the treatment of a wide range of illnesses, such as minimizing the adverse effects of immunosuppressive therapy with steroids.


Study of the effectiveness of glucosamine and chondroitin sulfate, marine based fatty acid compounds (PCSO-524 and EAB-277), and carprofen for the treatment of dogs with hip osteoarthritis: A prospective, block-randomized, double-blinded, placebo-controlled clinical trial



Canine tracheal collapse is a progressive disease in small breed dogs resulting from chronic inflammation of the tracheal mucosal lining. Polyunsaturated fatty acid EAB-277® is one of the nutraceuticals that can alleviate inflammation and oxidative stress. Heart rate variability (HRV) is a prognostic tool related to sympathovagal balance and oxidative stress level, which is widely used with cardiorespiratory diseases. However, the effect of EAB-277® on HRV in tracheal collapse dogs has rarely been investigated.
Introduction: Glucosamine hydrochloride and chondroitin sulfate are commonly used in dogs with OA, but evidence around efficacy is mixed. This study evaluated the effectiveness of glucosamine and chondroitin sulfate, marine based fatty acid compounds (PCSO-524 and EAB-277), and carprofen for the alleviation of canine hip OA pain. This was a prospective, block-randomized, double-blinded, placebo-controlled clinical trial.
Methods: Seventy-five owned pet dogs with hip OA were assigned randomly into five treatment groups: PCSO-524, Glucosamine and chondroitin sulfate, EAB-277, carprofen, and Placebo (sunflower oil). Peak vertical force (PVF) and subjective orthopedic assessment scores (OAS) were evaluated before treatment (week 0), and at weeks 2, 4, and 6 during treatment.
Results: At week 2, the carprofen group showed a significant increase in PVF (3.14 ± 5.33; mean ± SD). After 4 weeks, the increases in PVF of the PCSO-524 (3.90 ± 3.52), EAB-277 (4.17 ± 4.94), and carprofen (3.08 ± 5.87) groups were significant, and significantly greater than placebo (0.08 ± 1.90) and glucosamine (−0.05 ± 6.34) groups. After 6 weeks, the change of PVF in the PCSO-524 (4.14 ± 4.65), EAB-277 (4.45 ± 4.23), and carprofen (4.21 ± 6.52) groups were significant and significantly higher than the placebo group (−0.33 ± 3.65). The change in PVF in the glucosamine group (1.08 ± 5.49) lay between the placebo group and the other treatment groups. The OAS did not show any significant change in any group.
Discussion: PCSO-524 and EAB-277, but not glucosamine/chondroitin, resulted in significant improvements in PVF from baseline after 4 weeks, and 6 weeks, and to a similar degree to that seen with carprofen.

Updated Study Topics
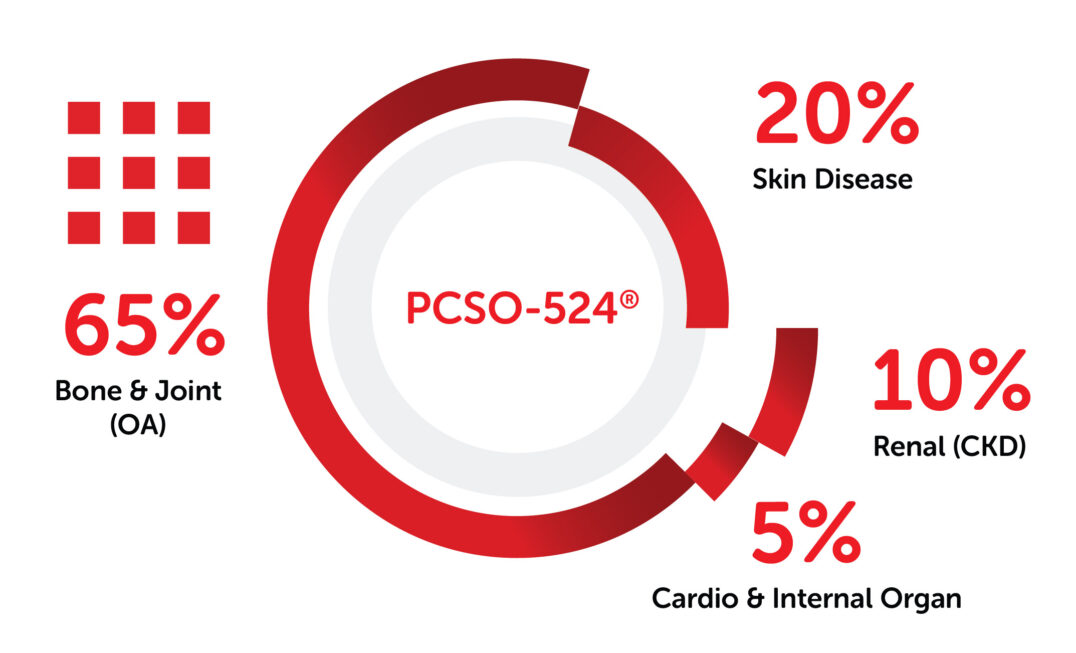
New researches and publications related to PCSO-524® and its result in clnical test submitted by veterinarians on the global conferences every year and the data keeps growing with more studies conducted

 Authors:
Authors: